Experimental setup to assess commercially available reference material... | Download Scientific Diagram

PDF) Analytical performance evaluation of a commercial next generation sequencing liquid biopsy platform using plasma ctDNA, reference standards, and synthetic serial dilution samples derived from normal plasma

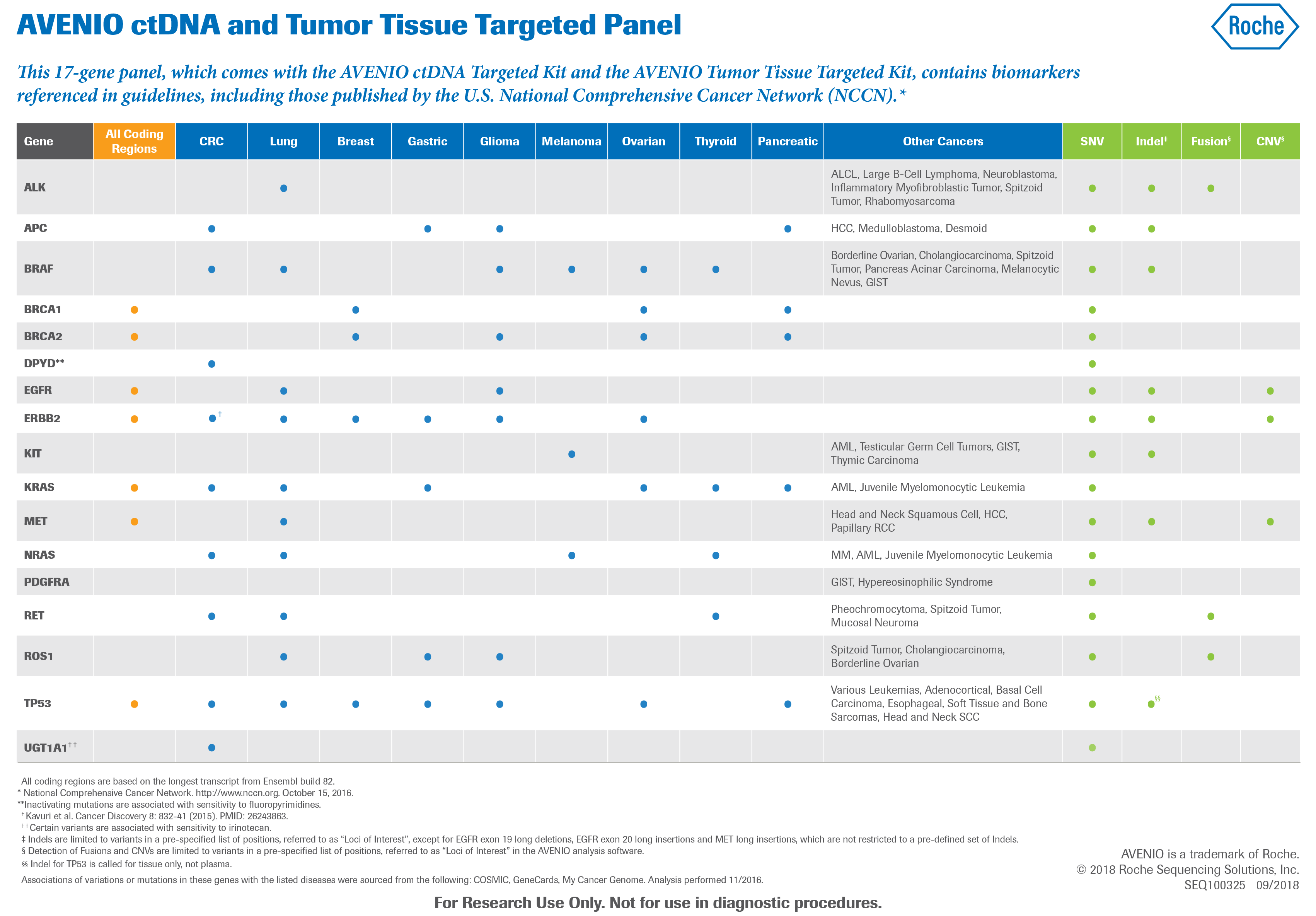
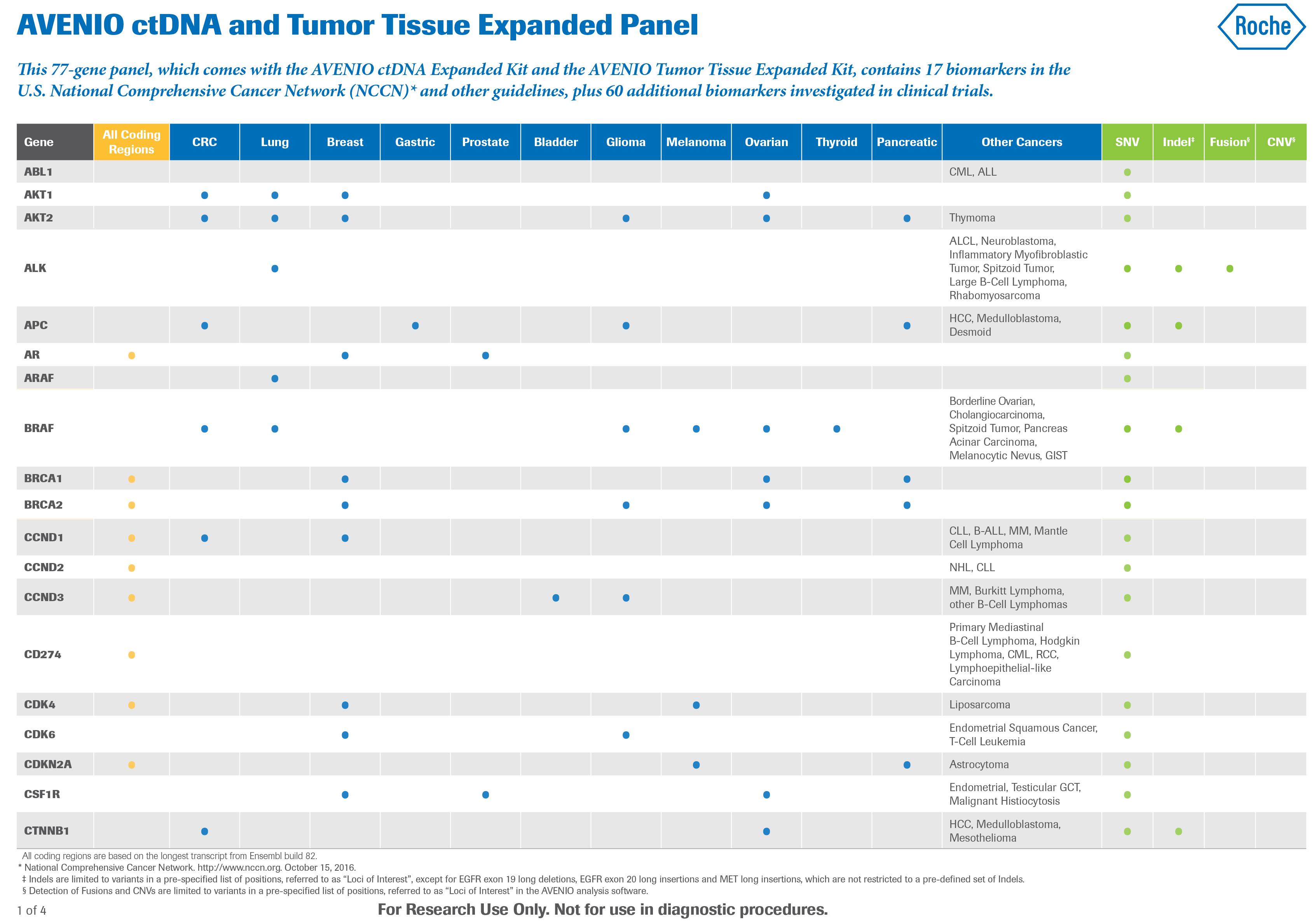
Kit di reagenti NGS - AVENIO - Roche Sequencing Solutions - da ricerca / per estrazione di DNA / per oncologia

Analytical performance evaluation of a commercial next generation sequencing liquid biopsy platform using plasma ctDNA, reference standards, and synthetic serial dilution samples derived from normal plasma. - Abstract - Europe PMC

A clinician's handbook for using ctDNA throughout the patient journey | Molecular Cancer | Full Text

AVENIO Tumor Tissue Surveillance Kit – Longitudinal Tumor Burden Monitoring - Roche Sequencing Store
Early Evaluation Site Experience with a Liquid Biopsy Kit designed for Next Generation Sequencing of Circulating Tumor DNA
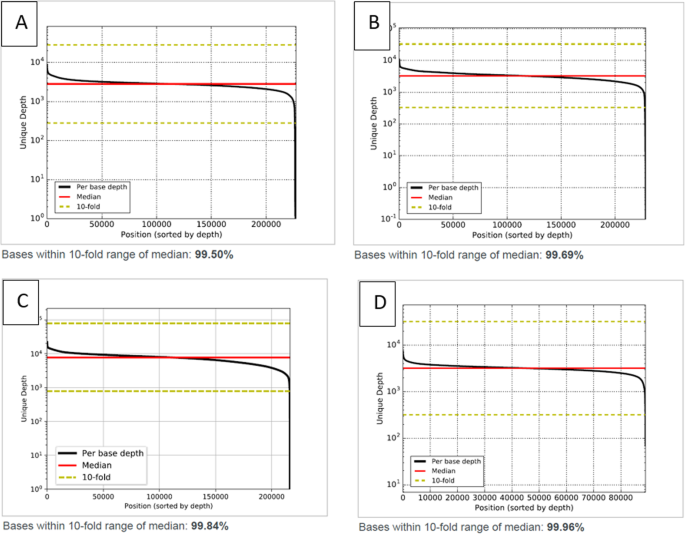
Analytical performance evaluation of a commercial next generation sequencing liquid biopsy platform using plasma ctDNA, reference standards, and synthetic serial dilution samples derived from normal plasma | BMC Cancer | Full Text